How to perform a titration for acid chemicals
Keeping on top of the chemistry in a car wash is critical to ensure that you are delivering a consistent quality to your customers, but also that you are not wasting product or at risk of causing vehicle damage from over dosing. In a touch free wash this is critically important, as you are relying on the performance of your soaps to deliver clean, shiny cars. The most common and simple titration to do is on your presoaks, as these are the highest use in volume and the most likely to affect your wash results.
This month we will show you how to do an Acid titration. The actual results will be dependent on the particular product you are using and you should talk to you chemical supplier to provide you with the test method and table for their products. We are often asked about using pH for testing chemical strength. While pH has a role to play in the car wash environment, it is not an accurate measure of the chemical strength. A titration will measure the amount of “free alkalinity” available to provide cleaning power, and is therefore a much more accurate test of strength and performance.
How to Perform a Titration for Acid (Low pH) Chemicals
Step One: Rinse the sample vial with the test solution.
Step Two: Fill the test tube to the 10 ml mark with the solution to be tested.

Step Three: Add 3-5 drops of Indicator P (#014) and mix. Solution will remain clear in colour (unchanged).

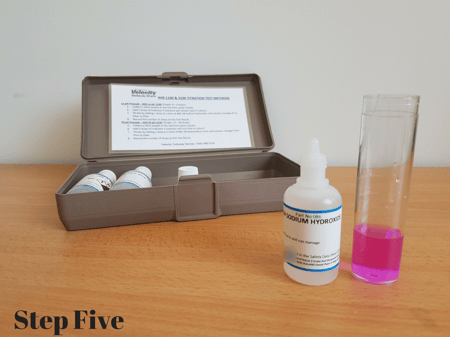
Step Four: Titrate with reagent 1.0N Sodium Hydroxide (#086) one drop at a time, counting and mixing between each drop until the solution changes from colourless to pink.

Step Five: Record number of drops required to reach pink end point, this is your final titration result.
Step Six: Consult Product Specification Sheet (PSR) to calculate product concentration. PSR’s for all Velocity Vehicle Care products are available for download at www.velocityvehiclecare.com

Click Here to Download – How to Perform an Acid Titration PDF















